42 Mo (Molybdenum)

The metal is silvery white, very hard, But is softer and more ductile than tungsten.
It has a high elastic modulus, and after “Mo” only tungsten and tantalum are the more readily available metals, which have higher melting points.
Molybdenum is attacked slowly by acids.

Identity
CAS Number: CAS7440-98-7
CID Number: CID23932
DOT Hazard Class: 4.1
DOT Number: 3089
RTECS Number: RTECSQA4680000
CONTENT INDEX
Basic Properties of Molybdenum
Pronunciation: Ma-lib-den-an
Appearance: Gray Metallic
Mass Number: 96
Standard Atomic weight: 95.95 g/mol
Atomic number (Z): 42
Electrons: 42
Protons: 42
Neutrons: 54
Period: 5
Group: 6
Block: d
Element category: Transition metal
Electrons per shell: K2, L8, M18, N13, O1
Electron configuration: 1s22s22p63s23p63d104s24p64d55s1

Thermal Properties of Molybdenum
Phase: Solid
Melting point: 2896 K (2623 oC, 4753 oF)
Boiling point: 4912 K (4639 oC, 8382 oF)
Debye temperature: 450 K (176.85 oC, 350.33 oF)
Fusion heat: 37.48 kJ/mol
Vaporization heat: 598 kJ/mol
Specific heat: 251 J/(kg K)
Molar heat capacity: 24.06 J/(mol.K)
Thermal diffusivity: 54.3 mm2/s
Thermal expansion: 4.8 μm/(m∙K)
Thermal conductivity: 138 W/(m∙K)
Electrical properties of Molybdenum
Electrical conductivity: 20×106 S/m
A Electrical resistivity: 53.4 nΩ∙m
A Electrical type: Conductor
Critical point (Superconducting point): 0.915 K (-272.23 oC, -458.01 oF)
Magnetic Properties of Molybdenum
A Magnetic type: Paramagnetic
Magnetic susceptibility (xmol): +89×10-6 cm3/mol
Volume magnetic susceptibility: 0.0001203
Mass magnetic susceptibility: 11.7×10-9 m3/kg
Molar magnetic susceptibility: 1.122×10-9 m3/mol
Physical Properties of Molybdenum
Density: 10.28 g/cm3 (In solid) 9.33 g/cm3 (In Liquid at MP)
Molar volume: 0.000009334 m3/mol
Young’s modulus: 329 GPa
Shear modulus: 126 GPa
Mohs Hardness: 5.5
Bulk modulus: 230 GPa
Poisson ratio: 0.31
Vicker hardness: 1400-2740 MPa
Brinell hardness: 1370-2500 MPa
Sound Speed: 5400 m/s
Atomic Properties of Molybdenum
Oxidation states: 6, 5, 4, 3, 2, 1, -1, -2, -4
Valence Electrons: 4d5 5s1
Ion charge: Mo6+
The ionization potential of an atom: 7.35
Ionization energies: 1st: 684.3 kJ.mol 2nd: 1560 kJ/mol 3rd: 2618 kJ/mol
Ionic radius: 65 pm
Atomic radius: empirical: 139 pm
Van der Waals: 209 pm
Covalent radius: 154±5 pm
Filling Orbital: 4d5
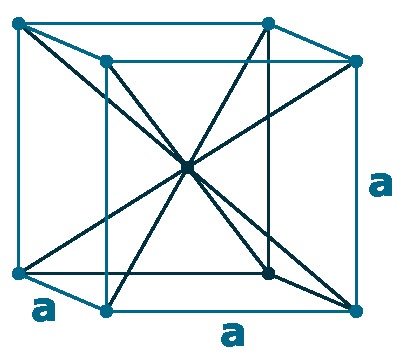
Crystal structure: Body-centered cubic
Lattice angles: π/2, π/2, π/2
Lattice constant: 314.7, 314.7, 314.7 pm
Grid parameters: a=3.147 Å
Space Group Name: lm_3m
Space Group Number: 229
Reactivity of Molybdenum
Electronegativity: pauling scale: 2.16
Valence: +6
Electron affinity: 71.9 kJ/mol
Nuclear Properties of Molybdenum
Half Life: Stable (Infinity)
Lifetime: Stable (Infinity)
Quantum Number: 7S3
Neutron cross section (Brans): 2.6
Neutron Mass Absorption: 0.0009
Isotopes: 92Mo 93Mo 94Mo 95Mo 96Mo 97Mo 98Mo 99Mo 100Mo
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 92Mo | 14.65 | 91.907 | Stable |
| 93Mo | Syn | – | 4×103 y |
| 94Mo | 9.19 | 93.905 | Stable |
| 95Mo | 15.87 | 94.906 | Stable |
| 96Mo | 16.67 | 95.905 | Stable |
| 97Mo | 9.58 | 96.906 | Stable |
| 98Mo | 24.29 | 97.905 | Stable |
| 99Mo | Syn | – | 65.94 h |
| 100Mo | 9.74 | 99.907 | 7.8×1018 y |
Chemical Reactions of Molybdenum
It doesn’t react with air at room temperature, But If heated it to the elevated temperature (red Heat) It forms:
2 Mo (s) + 3 O2 (g) → 2 MoO3 (s) (trioxide molybdenum (VI) oxide)
At room temperature, molybdenum does not react with water.
Reacts with Halogens:
At room temperature, react with fluorine:
Mo (s) + 3 F2 (g) → MoF6 (l) [colourless] (molybdenum (VI) fluoride)
Under controlled conditions, react with chlorine:
2 Mo (s) + 5 Cl2 (g) → 2 MoCl5 (s) [black] (molybdenum (V) chloride)
Reacts with sulfide:
Mo(VI) is precipated by sulfide in 0.4 M hydrochloric acid (HCl)
MoO42- (aq) + 3 S2- (aq) + 8 H+ (aq) ⇌ MoS3 (s) [brown/black] + 4 H2O (l)
The precipitate can be dissolved by sodium di sulfide (Na2S2):
2MoS3 (s) + S22- (aq) ⇌ 2 MoS42-
Mo (VI) as ammonium molybdate is precipitated by hydrogen sulfide (H2S) in the presence of ammonia (NH3):
MoO42- (aq) + 4 H2S (aq) + 2 NH4+ (aq) ⇌ (NH4)2MoS4 (s) [red] + 4 H2O (l)
Nitrogenases catalyze the production of Ammonia from atmospheric nitrogen:
N2 + 8 H+ + 8 e– + 16 ATP + 16 H2O → 2 NH3 + H2 + 16 ADP + 16 Pi
Production:
molybdenite processing, Firstly ore is roated at 700 oC, It gives gaseous sulfur dioxide (SO2) and the molybdenum(VI) oxide:
2 MoS2 + 7 O2 → 2 MoO3 + 4 SO2
oxidized ore, usually extracted with aqueous ammonia, and give ammonium molybdate:
MoO3 + 2 NH3 (aq) + H2O → (NH4)2(MoO4)
Ammonium molybdate converts to ammonium di molybdate ((NH4)2Mo2O7), which is isolated as a solid. Heating this solid, ammonium dimolybdate is decomposes to molybdenum trioxide:
(NH4)2Mo2O7 → 2 MoO3 + 2 NH3 + H2O
Finally, Metallic molybdenum is produced by reduction of the oxide with hydrogen:
MoO3 + 3 H2 → Mo + 3 H2O
Molybdenum History
Naming: Greek: molubdos (lead).
Discovery: Carl Wilhelm Scheele (1778) in Uppsala Sweden.
First isolation: Peter Jacob Hjelm (1781)
Molybdenum Uses
Molybdenum has a very high melting point so it is produced and sold as a grey powder.
Many molybdenum items are formed by compressing the powder at a very high pressure.
A Molybdenum is a valuable alloying agent, that is used in steel alloys to increase hardness, strength, electrical conductivity and resistance to corrosion and wear.
Almost all ultra-high strength steels with minimum yield points up to 300,000 psi contain molybdenum in amounts of 0.25 to 8%.
‘moly steel’ alloys are used in parts of engines.
It is used in certain nickel-based alloys, such as the “Hastelloys(R)” which are heat-resistant and corrosion-resistant to chemical solutions.
A Molybdenum sulfide is used as a lubricant additive, especially at high temperatures where oils would decompose.
The metal has recent use as electrodes for electrically heated glass furnaces and forehearths.
It is also used in nuclear energy applications and for missile and aircraft parts.
Use of molybdenum as a catalysts in the refining of petroleum.
It has found applications as a filament material in electronic and electrical applications
Biological role
Molybdenum and its compounds are highly toxic at larger doses, and it is an essential element for animals and plants, too much molybdenum causes fetal deformities.
At least 50 different enzymes used by plants and animals contain molybdenum.
One of these is nitrogenase, which is found in nitrogen-fixing bacteria that make nitrogen from the air.
Leguminous plants have root nodules that contain these nitrogen-fixing bacteria.
Molybdate is transported in the body as MoO42−.
Abundance of Molybdenum
The main molybdenum ore is molybdenite (molybdenum di sulfide, MoS2), and can be found in wulfenite (PbMoO4).
It is processed by roasting to form molybdenum oxide, and then reducing to the metal by the hydrogen reduction of purified molybdic trioxide or ammonium molybdate.
Molybdenum is also obtained as a by-product of copper and tungsten mining operations.


World wide production is around 2,50,000 tons per year
5×10-7% (In Universe)
12×10-5% (In Meteorites)
9×10-7% (In Sun)
0.00011% (In Earth’s Crust)
9.9×10-7% (In Oceans)
0.00001% (In Humans)
World’s Top 3 producers of Molybdenum
1) China
2) USA
3) Chile
World’s Top 3 Reserve holders of Molybdenum
1) China
2) USA
3) Chile
#Molybdenum


