18 Ar (Argon Element)

Argon is a colourless & odorless element in both liquid & gaseous forms, and considered to be a very inert gas.
It has the same solubility as oxygen, and 2.5 times as soluble in water as nitrogen.


Identity
CAS Number: CAS7440-37-1
CID Number: CID23968
DOT Hazard Class: 2.2
DOT Number: 1951
RTECS Number: RTECSCF2300000
CONTENT INDEX
Basic Properties of Argon
Pronunciation: Ar-gon
Appearance: colourless gas exhibiting a violet/lilac glow when placed in an electric field
Mass Number: 40
Standard Atomic weight: 39.963, 39.792 g/mol
Atomic number (Z): 18
Electrons: 18
Protons: 18
Neutrons: 22
Period: 3
Group: 18
Block: p
Element category: Nobal gas
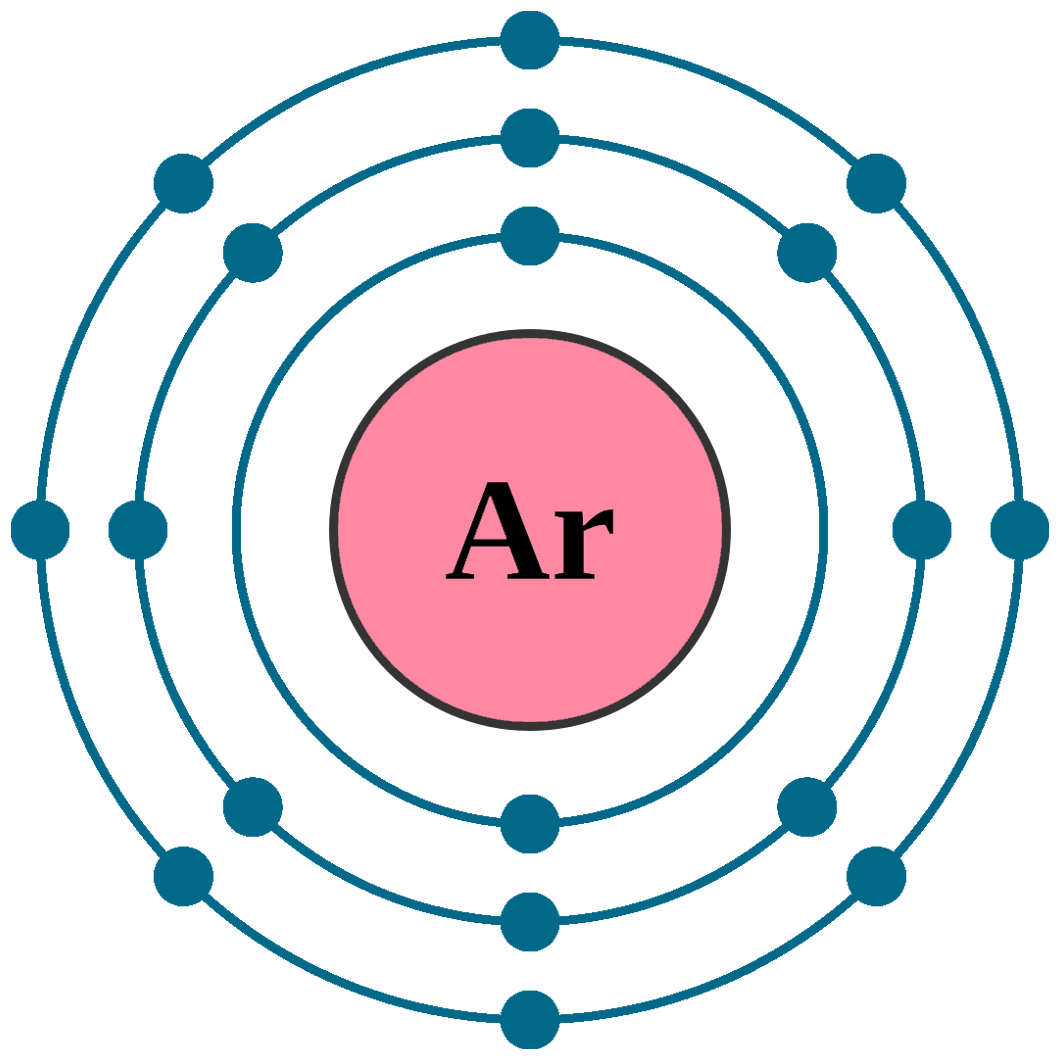
Electrons per shell: K2, L8, M8
Electron configuration: 1s22s22p63s23p6
Thermal Properties of Argon
Phase: Solid
Melting point: 83.81 K (-189.34 oC, -308.526 oF)
Boiling point: 87.302 K (-185.848 oC, -302.526 oF)
Debye temperature: 85 K (-188.15 oC, -306.67 oF)
Triple point temperature: 83.805 K (-189.345 oC, -308.821 oF)
Triple point pressure: 68.89 KPa (0.67 Atm)
Critical point temperature: 150.687 K (-122.463 oC, -188.4334 oF)
Critical point pressure: 4.863 MPa (48.29 Atm)
Fusion heat: 1.18 kJ/mol
Vaporization heat: 6.53 kJ/mol
Specific heat: 520 J/(kg K)
Molar heat capacity: 20.85 J/(mol.K)
Thermal conductivity: 17.72×10-3 W/(m∙K)
Magnetic Properties of Argon
A Magnetic type: Diamagnetic
Magnetic susceptibility (xmol): -19.6×10-6 cm3/mol
Volume magnetic susceptibility: -0.0000000107
Mass magnetic susceptibility: -6×10-9 m3/kg
Molar magnetic susceptibility: -0.24×10-9 m3/mol
Physical Properties of Argon
Density: 1.784 g/cm3 (at STP) 1.395 g/cm3 (In Liquid at B.P)
Molar volume: 0.022392 m3/mol
Sound Speed: 323 m/s
Atomic Properties of Argon
Oxidation states: 0
Valence Electrons: 3s2 3p6
Ionization potential of an atom: 15.68
Ionization energies: 1st: 1521 kJ.mol 2nd: 2666 kJ/mol 3rd: 3931 kJ/mol
Van der Waals: 188 Pm
Covalent radius: 106±10 pm
Filling Orbital: 3p6
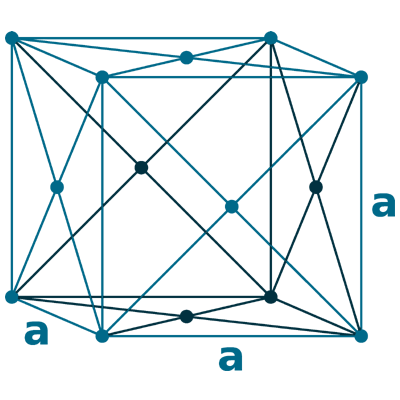
Crystal structure: Face centered cubic
Lattice angles: π/2, π/2, π/2
Lattice constant: 526.1, 526.1, 526.1 pm
Grid parameters: a=5.260 Å
Space Group Name: Fm_3m
Space Group Number: 225
Nuclear Properties of Argon
Half Life: Stable (Infinity)
Lifetime: Stable (Infinity)
Quantum Number: 1S0
Neutron cross section (Brans): 0.66
Neutron Mass Absorption: 0.00059
Isotopes: 36Ar 37Ar 38Ar 39Ar 40Ar 41Ar 42Ar
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 36Ar | 0.334 | 35.967 | Stable |
| 37Ar | Syn | – | 35 d |
| 38Ar | 0.062 | 37.964 | Stable |
| 39Ar | Trace | – | 269 y |
| 40Ar | 99.604 | 39.962 | Stable |
| 41Ar | Syn | – | 109.34 min |
| 42Ar | Syn | – | 32.9 y |
Chemical Reactions of Argon
Argon is inert gas, and doesn’t react with anyone.
Argon History
Naming: Greek: Argos (inactive).
Discovery & first isolation: Lord Rayleigh and William Ramsay (1894) in London & Bristol (England)
Argon Uses
Argon is often used there, where an inert (chemically inactive) atmosphere is needed.
It is used in the production of titanium & other reactive elements, and also used by welders in shielded arc welding to protect the weld area & in incandescent light bulbs to stop oxygen from corroding the filament.
It is used in fluorescent tubes & low-energy light bulbs, where low-energy light bulb often contains argon gas & mercury.
When the bulb is switched on, an electric discharge passes through the gas, which generating UV light, and the coating on the inside surface of the bulb is activated by the UV (Ultraviolet) light, which glows brightly.
It provides protective atmosphere for growing silicon & germanium crystals.
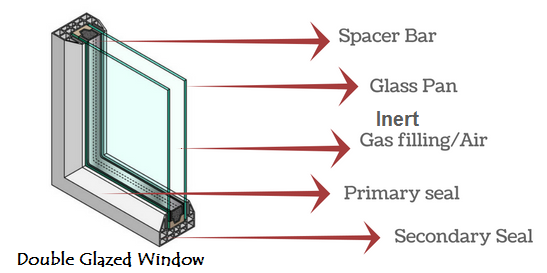
Double-glazed windows (two panes of glass) use argon to fill the space between the panes.
Argon gas is also used in the tyres of Luxury cars to protect the rubber & reduce road noise.
Argon has been used in number of applications, such as: ice coring, ground water dating, in technical SCUBA diving to inflate the drysuit, due to its nonreactive, heat isolating effect.
It is also used in scientific research for neutron experiments & dark matter searches.
Biological role of Argon
It is non-toxic, and 38% denser than air, therefore it considered a dangerous Asphyxiant in closed areas.
After inhalation, It could be a serious risk of suffocation.
Because of its odorless, colourless, & tasteless form, it is difficult to detect.
Abundance of Argon
Argon is the 3rd most abundant atmospheric gas, and it makes up 0.94% of the Earth’s atmosphere.
Commercially, It is obtained by fractional distillation of liquid air in a cryogenic air separation unit.
Annual world wide production of Argon is around 7,50,000 tons.
0.02% (In Universe)
0.007% (In Sun)
0.00015% (In Earth’s Crust)
0.000045% (In Oceans)
Argon Price
Pure Liquid Argon price is around $5-$10 per KG (KiloGram)
#Argon


