Metalloids On The Periodic Table
CONTENT INDEX
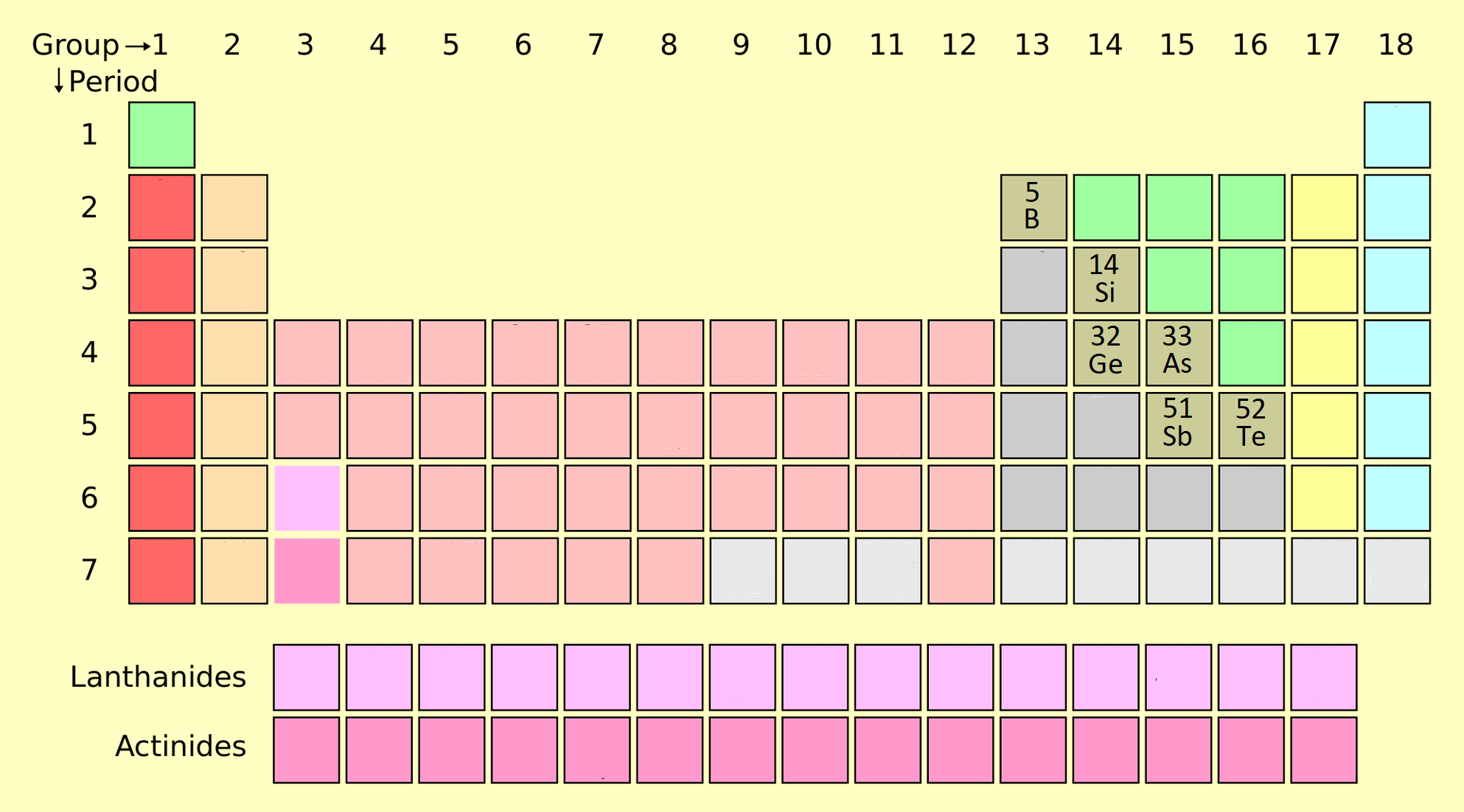
About Metalloids
Metalloids are actually those materials that have properties could belong to either metals or non-metals. Their properties somewhere similar to metals and non-metals both. Silicon is known as the best example of metalloids. These are located between Post-Transition metals and Non-metals on the periodic table. Many other elements are also included in metalloids that we will discuss further. We will also discuss about their properties and characteristics. So, firstly we will discuss about their properties. After that we will discuss about their characteristics and uses in our daily life.
Properties of Metalloids
- They are generally solids like metals, and brittle like non metals.
- Their ability to conduct heat is in between metals and non-metals.
- They conduct electricity at higher temperature.
- Some metalloids such as silicon and germanium can act as electrical conductors under the specific conditions thus, they are called semiconductors.
- Silicon is the best example of metalloid, But silicon is not the only metalloid. The material other than silicon are officially included.
- Metalloid groups are Silicon (Si), Boron (Br), Germanium (Ge), Arsenic (As), Antimony (Sb) and Tellurium (Te).
- Arsenic (As) is best known as poisonous substance.
- They are also known as semi-metals.
- All the metalloids are found in solid state at room temperature.
- Alloy are the type of metalloid which are formed by mixing two or more metals together. For example- brass is an alloy which is made from Copper and Zinc metals.
- Similar to metals, metalloids can form alloys with other metals.
- Some metalloids have lustrous look. For example – silicon is shiny matter. It is neither malleable nor ductile. It is much poorer conductor of heat and electricity than the metals.
Characteristics of Metalloids
1) Occurrence: it can be found in the periodic table in diameters from boron (B) to statin (At). It is divided into columns 13, 14, 15, 16 and 17. The table is divided into two elements, the elements in the middle on the right are non-metallic and the elements on the left are metallic.
2) Form and coloration: The metalloids are varied in their shape and color. They can be transparent or opaque, and many have more than one descriptive state that varies in width depending on their molecular structure. For example, arsenic can be gray, yellow, or black, depending on the allotrope. Silicon can be presented as both a shiny solid crystal and an amorphous brown powder.
3) Electric conduction: Most metals are semiconductors. They only conduct electricity in one direction instead of being fully conductive, which is what sets them apart from metal elements. However, they are much better motors than non-metallic insulating elements, so they are widely used in the power generation industry.
4) Thermal conduction: like electricity, metals conduct thermal energy much better than non-metals without reaching the high conductivity of metals. In general, they are not considered good conductors of heat.
5) Reactivity: Metals react differently due to their intermediate state depending on the presence of the metal element and then react as a non-metallic and then react as a metal. They are related to different elements depending on the element, which is why they are usually found with minerals such as Uranium, Lead, Sulphur etc.
6) Toxicity: although some metals, such as arsenic, are essential for the formation of biological molecules and are found in living organisms. May pose a health risk. Boron or arsenic themselves can be somewhat toxic, and polonium is a highly toxic, radioactive element that gives off harmful alpha particles.
7) States of aggregation: most metalloids are usually shown in the solid state at room temperature either in the form of crystals such as arsenic or opaque and brittle blocks such as germanium. However, some have curious properties in terms of phase change. For example – arsenic passes directly from solid to gas sublimation in the presence of heat and at normal atmospheric pressures.
8) Atomic properties: Metalloids are very diverse in their atomic characterization. There are some with great atomic weight. Such as polonium, its atomic weight is 127.6 and other elements such as boron weight has 10.81. And silicon has 28.08. Same happens with their densities ranging from 2.37 gram per centimeter cube (Boron) to 9.32 gram per centimeter cube (polonium).
9) Abundance: Some metalloids are abundant in the earth’s crust, such as B. Silicon, which forms several compounds called silicates or arsenic. Boron, on the other hand, is abundant, but as part of a mineral called Borax or sodium borate (Na2B4O710H2O or Na2[B4O5(OH)4]·8H2O), as it does not occur naturally in a free or pure form. Polonium, on the other hand, is very rare and only occurs as part of some uranium minerals. Antimony occurs in very small percentages in the earth’s crust.
10) Utility: Most semi-metallic materials are used to make electronic and semiconductor components such as rectifiers and transistors, even in the case of silicon chips and microprocessors. However, their versatility makes them very versatile. For example, some isotopes of boron are useful in neutron capture in nuclear power plants, which also serve as mechanisms for regulating atomic reactions.
Uses of metalloids
- Silicon (SiO2)
- Silicon is a semiconductor and heavily used in electrical industries.
- It is also used in manufacture of water proof material called silicone such as bags, umbrellas, raincoats.
- Sand is the main ingredient in manufacturing of glass.
- Other metalloids such as boron, gallium is also used in electrical devices.
- Boron used in fireworks and as a fuel for ignition in rocket.
- Germanium is used as semiconductor and to form alloys and phosphor in fluorescent lamps.
- Antimony is used to make semiconductor devices. And it is also used in printing press as type metal (Pb, Sb, Sn). It alloyed with lead and used in batteries.
Metalloids Interesting Facts:
- The most common metal in the earth’s crust is silicon, the second most common element overall (oxygen is more common).
- The least natural mineral is tellurium. Metals are valuable in the electronics industry. For example, silicon is used to manufacture telephone and computer chips.
- Arsenic and polonium semimetals are highly toxic. Antimony and tellurium are mainly used in metal alloys to add desirable properties.
We have discussed above about metalloids and their properties or characteristics. We have also discussed about their uses in our daily life. So, now I hope you get all the information regarding this topic.




I really enjoyed this post on metalloids! The detailed breakdown of their properties and uses was informative and eye-opening. It’s fascinating how these elements play such a crucial role in technology. I especially appreciated the section about their applications in semiconductors. Looking forward to more posts like this!